The interaction between drugs and receptors forms the foundation of pharmacology, determining how medications influence biological systems at a molecular scale. Understanding these interactions is essential for grasping drug effectiveness, specificity, and potential side effects. This article explores the molecular mechanisms behind drug-receptor interactions and their significance in drug development.
1. Introduction
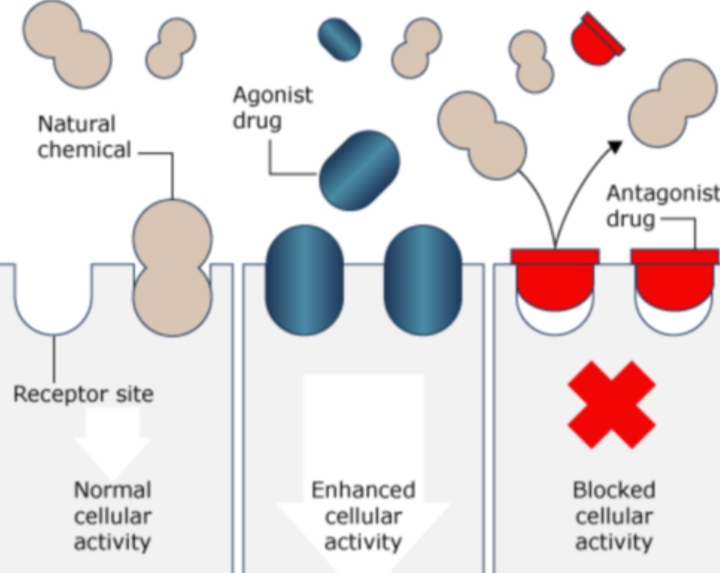
Receptors are specialized proteins found on cell membranes or inside cells that recognize and respond to natural signaling molecules such as hormones, neurotransmitters, or cytokines. Drugs can mimic or inhibit these natural ligands to modify physiological processes. The selectivity and strength of drug binding largely dictate therapeutic outcomes and side-effect profiles.
2. Molecular Interactions in Drug Binding
At the molecular level, drugs interact with receptors through non-covalent forces, including:
- Hydrogen bonds: Connections formed via hydrogen atoms contribute to specific binding.
- Ionic interactions: Electrostatic attractions between charged groups stabilize the drug-receptor complex.
- Hydrophobic interactions: Non-polar regions of drugs and receptors cluster together, increasing binding affinity.
- Van der Waals forces: Weak, transient interactions enhance the overall fit between drug and receptor.
These interactions determine whether a drug functions as an agonist, antagonist, or allosteric modulator.
3. Types of Receptors
Receptors are categorized based on their structure and mechanism:
- G Protein-Coupled Receptors (GPCRs): Span the cell membrane seven times and signal through G proteins; a major target for many drugs.
- Ion Channel Receptors: Control ion flow across membranes and can be activated or blocked by drugs.
- Enzyme-Linked Receptors: Possess intrinsic enzymatic activity, like tyrosine kinases, mediating growth and differentiation.
- Intracellular Receptors: Located inside the cell, these respond to lipophilic drugs such as steroids and influence gene transcription.
4. Binding Kinetics and Affinity
Drug-receptor interactions are dynamic, governed by the rates of binding (association) and release (dissociation). The equilibrium dissociation constant (K_d) measures a drug’s binding affinity:
[
K_d = \frac{k_{off}}{k_{on}}
]
A lower K_d indicates stronger binding. However, strong binding does not always equate to strong biological effects; a drug can attach tightly without triggering receptor activation.
5. Agonists, Antagonists, and Allosteric Modulators
- Agonists activate receptors by stabilizing their active conformation.
- Antagonists bind without activating, blocking natural ligands.
- Partial agonists elicit weaker responses even when fully bound.
- Allosteric modulators attach to alternate sites on the receptor, modifying the effect of the natural ligand.
6. Structural Insights
Techniques like X-ray crystallography, NMR, and cryo-electron microscopy reveal receptor structures in complex with drugs. These structural insights guide rational drug design, allowing development of molecules with enhanced specificity, reduced side effects, and better pharmacological profiles. For instance, designing selective β2-adrenergic receptor agonists has greatly improved asthma treatment.
7. Clinical Relevance
Understanding drug-receptor interactions at the molecular level helps:
- Enhance drug selectivity: Reducing unintended effects by targeting specific receptors.
- Address resistance: Mutations in receptor sites can alter drug effectiveness.
- Develop multi target drugs: Designing medications that act on multiple receptors to treat complex diseases.
Conclusion
A molecular perspective on drug-receptor interactions is crucial for understanding drug action. Insights into binding forces and receptor structures provide a foundation for rational drug design, enabling the creation of more precise and effective therapeutics. Advances in structural biology and molecular modeling continue to refine our approach to drug development.
